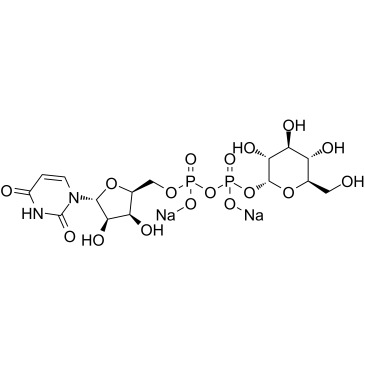
[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl] phosphate
We are professional UDP-Glc manufacturer and Uridine 5′-Diphosphoglucose Disodium Salt supplier in China, We offer quality disodium,[[(2R,3S,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-oxidophosphoryl] [(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl] phosphate you can fully trust, also we have India factory and producer of Uridine 5′-Diphosphoglucose Disodium Salt,Pls send inquiry of Uridine 5′-Diphosphoglucose Disodium Salt to info@nbinno.com if you have any interests, thank you!
Related News: Biogen last month revived its plans to seek U.S. approval for its aducanumab treatment after announcing in March that it would terminate two large clinical trials for the drug. But some analysts believed FDA approval is highly unlikely.2-Amino-4-nitrophenol CAS:99-57-0 In 2016, in order to promote the innovative development, transformation and upgrading of the pharmaceutical industry, the Development and Reform Commission led the compilation of the “Guiding Opinions on Promoting the Healthy Development of the Pharmaceutical Industry”, which put forward requirements for all aspects of the medical industry and specifically proposed support for the field of chemical raw materials. .1-Chloro-4-fluorobutane CAS:462-73-7 The term active pharmaceutical ingredient may refer to an active chemical within an FDA-regulated drug, or API might mean the entire drug with its active and inactive ingredients.non-8-enoic acid Immediately thereafter, the iLet begins controlling blood-sugar levels automatically, without requiring the user to count carbohydrates, set insulin delivery rates, or deliver bolus insulin for meals or corrections.Under the terms of this agreement, Inceptua will support Onconova through the pre-approval provision of intravenous rigosertib initially into a number of countries including: Australia, Denmark, Finland, France, Ireland, Italy, the Netherlands, Portugal, South Africa, Spain, and the UK.


